BECE Past Questions & Answers – 1998 (SCIENCE)
August 1998
SCIENCE
SECTIONB ESSAY
1hour
[60 marks]
Answerthreequestionsonlyfrom this section.
Illustrate your answers wherever possible, with large, clear and fully labelled diagrams. Creditwill be given for clarity of expression andorderlypresentation ofmaterial.
All questions carry equal marks
1. (a) Statetwo differences between aplanet andastar.
(b) (i) What is surfacetension.
(c) (ii)
(i) Stateonewayin which surfacetension can beuseful to man.
Giveone example eachof arhizome, a corm,astem tuber, abulb and
asucker.
(ii) Statetwo reasons whyarhizome, a corm and astem tuber are consideredas stems.
(d) Name the compounds formed when the following elements combine:
(i) (ii) (iii) (iv) zinc and oxygen calcium and chlorine sodium and bromine potassiumand iodine
2.
(a)
(i)
What is a fertile soil?
(ii) Name four of themostimportant nutrients in mineral saltswhich plants useto form food.
(b)
(i)
Definedensityofasubstance.
(ii) (iii) Describebrieflyhowyouwould determinethe densityof an irregularpieceofstone.
A boxwith volume 1200.0 m3is filled with sand to the brim.If thedensityofthe sand is
1.95 kgm-3, calculatethemass ofthe sand contained in thebox.
(c)
(i)
Define anatom.
(ii) Whyare atoms electricallyneutral?
3. (a) Explain the following
(i) Cracklingnoisesareheard from a metal sheet roofwhen the weatherbecomes veryhot. (ii) Railwaytracks arelaid with gaps in between theirends.
(iii) A fittingmechanicwashes his greasyhandswith petrol and not with water.
(b) What observation willbemadeif apieceof bluelitmus paper is dropped ineach of the following:
(i) sodium hydroxide solution
(ii) dilute hydrochloricacid
(iii) lime juice
(iv) distilled water
(v) rain water
(c) State anythreeways inwhich constipation could be avoided.
(d) What arethe end products of thedigestion of eachof the following foodsubstances? (i) Carbohydrates
(ii) Proteins
(iii) fats
4. (a) State four ways bywhichplants areableto protectthemselves againstanimals.
(b) (i) Definepollination.
(ii) Listfourfeatures of insect pollinated flowers.
(c) An atomhas threeprotons and threeneutrons.
(i) Howmanyelectrons aretherein this atom?
(ii) Drawalabeled diagram to show the arrangementof allthe particles in theatom.
(d) A bodyof mass 12.0 kgis placed on an orangetree20.0 m abovetheground
(i) Calculate its potential energywith respect to theground
(ii) Ifthe bodyis released from thisheight, what will be its kinetic energyjustbeforeithits the ground.
[g=10 ms-2]
CLICK TO VIEW ANSWERS TO PART 2
SCIENCESectionB
SOLUTIONS
ESSAY
1. (a) Differences
Planet Star
Does not producelight Produces light
Madeup ofsolid and liquid materials
Madeup ofburning gases
Revolves aroundastar (sun) in a
specific orbit Does not move / stationary
(ie, relative to the planets)
[anytwo]
(b) (i) Surface tension
A condition existingat thesurfaceofaliquid, resemblingastretched elasticskin
Or: the propertyofaliquid that makes its surfacebehavelikeastretched elasticskin
(ii) Ways inwhichsurface tensionis useful to man
Surfacetension propertyof waterensures that waterdoes not leak throughraincoats, umbrellas, tents, canopies or other materials madeof canvas.
(c) (i) EXAMPLE Rhizome – ginger
Corm – cocoyam
Stem tuber – yam Bulb – onion Sucker – banana
(d) COMPOUNDSFORMED
(i) zincandoxygen – Zinc oxide or ZnO
(ii) (iii) Calciumandchlorine
Sodiumand bromine – Calcium chloride
– Sodiumbromide or or CaCl2
NaBr
(iv) Potassiumandiodine – Potassiumiodide or KI
2. (a) (i) Fertile soil
(ii) Soilthat is rich in allthesoilnutrients required forhealthyplant growth
Most important nutrients inmineral salts
Phosphorus, Nitrogen, Calcium, Magnesium, Potassium, Sulphur, Iron
[anyfour]
(b)
(i)
Density ofa substance
Themass per unit volume ofthe substance
Or: Density = mass / volume
(ii)
Finding density ofanirregular pieceofstone
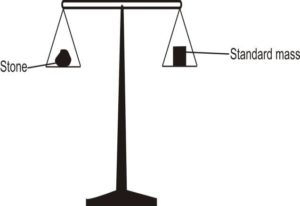
I. Measurethe mass(M)ofthe stone usingabeam balance II. Fill a measuringcylinderwith waterto a suitablevolume. III. Record the volume (V1)
IV. Tie athread / string around thestone andgentlylower itinto the waterin the
cylinder.
V. Record the new volume (V2)
VI. Find differencein volumes (V2– V1)
VII. Volumeofthestone = (V2– V1)
VIII.Findthe ratio M/ (V2– V1)= Density ofstone
(iii) Mass ofsand = Density × volume
= 1.95 kgm-3 × 1200.0 m3
= 3900kg
(c) (i) Atom
Thesmallest part of anelement that can takepart in a chemical reaction
Or:
Thesmallest portion into which an element can bedivided and still retain its properties
(ii) Why atoms are electrically neutral
Thenumbers of protons and electrons inan atomareequal. Thepositivelycharged protons neutralizethe negativelycharged electrons, hence, makingtheatomelectrically neutral
3. (a) Explanation
(i) Crackling noisesheard from ametal sheet roof when hot
Theheat causes themetal sheet roofto expand. The expansion causes thebendingof the metal sheet if thereis not enough space. Thebendingcauses thecracklingnoise
(ii) Railway tracks arelaidwith gaps in betweentheir ends.
Thegaps provideallowance forthe expansion of the railwaytracks, so thattheydo not bend duringhot periods,when expansion occurs.
(iii) A fitting mechanic washes his greasy handswithpetrol and notwithwater Greaseis solublein petrol, butnot solublein water. Hence, the petrol dissolves the greaseon his hands.
(b) Observation madewhen bluelitmus paperis droppedin:
(i) sodium hydroxide solution – colour remains blue
(ii) dilute hydrochloricacid – colour changes to red
(iii) lime juice – colour changes to red
(iv) distilled water – colour changes to purple
(v) rain water – colour changes to red /pink
(c) Ways ofreducing constipation
Drinking alotof water/liquids
Eatingfruits and vegetables regularly
Eatingfoods thatcontainhigh amounts of fiber, such as wheat, barley,etc
Gettingenough sleep
Exercisingregularly/ daily
(d) End products of digestionof:
(i) Carbohydrates – glucose
(ii) Proteins – amino acids
[anythree]
(iii) fats – fattyacids andglycerol
4. (a) Ways by which plants protect themselves against animals
Unpleasant smellfrom certain plants makes themrepulsiveto animals.
Unpleasant taste ofcertain plants makes themundesirable to taste/ eat.
Sharp thorns / spines of certain plants makethem difficult to touch.
Poisonous parts of certain plants can causeirritation / pain / infection / deathof animals.
Stickysecretions or partsof certain plants makes themuncomfortableto touch
[anyfour]
(b) (i) Pollination
Thetransfer ofpollengrains from the antherofa flower to thestigmaof another flower ofthe same species
(ii)
Features ofinsect pollinated flowers
Petals arebrightlycoloured
Larger sizeofflowers
Nectarpresent
Attractivescent
Relativelyfewer but larger pollengrains
[anyfour]
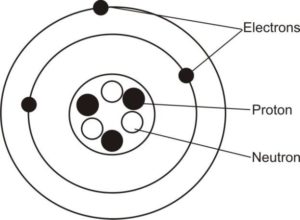
(c) Atomwith 3 protons and3 neutrons
(i) 3 electrons
(ii) Labeled diagram
(d) (i) Potential energy = mass ×height × acceleration duegravity
= 12 kg×20 m×10 ms-2
= 2400 kgm2s-2 or 2400 J
(ii) Kinetic energyjustbeforeithits the ground= 2400 J
