BECE Past Questions & Answers – 2004 (SCIENCE)
SECTION B
1 hour15 minutes
ESSAY [75marks]
This section is in two parts,I and II
Answer Question1 of Part I and any other three questions in Part II.
Credit will be given for clarity of expression and orderly presentation of material
PART I
[30 marks]
(Compulsory – Answer all of Question 1)
1. (a) Study the simple circuit diagram shown below and answer the questions that follow
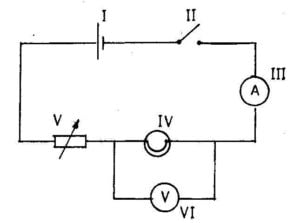
(i) Identify the components labelled I, II, III, IV, V, VI
(ii) Which component is used to close the circuit?
(iii) State the observation that will be made when the circuit is closed.
(iv) State the energy transformations that take place when the circuit is closed
(v) Give the name of the circuit connection between components IV, VI [10 marks]
(b) The diagram below is an illustration of an experimental set-up used for separating a mixture of water and alcohol. Study the diagram carefully and answer the questions that follow.

(i) State the method of separation shown in the set-up
(ii) Identify liquid Y, with reasons.
(iii) Give the functions of the thermometer and the water jacket X
(iv) What physical processes are involved in the method of separating the mixture?
(v) State the physical property which makes it possible to separate the water and alcohol [10 marks] (c) A student performed tests on food substances A,Band C and made the following observations
(i) Identify food substances A, B and C.
(ii) Give the products of digestion of A, B and C
(iii) In which parts of the alimentary canal does the digestion of each of food substances A, B and
C start?
(iv) In which part of the alimentary canal is food substance C absorbed after digestion? [10 marks]
PART II
[45 marks]
(Answer three questions from this part)
2. (a) (i) List four parts of a plant that are used for asexual reproduction
(ii) Give two reasons why sexual reproduction is more advantageous than asexual reproduction.
(b) (i) Explain why atoms are electrically neutral
(ii) Write the systematic names of the following substances:
(α) CuO,
(β) SO2
[5 marks]
(c) (i) What is dispersion of light?
(ii) Draw and label a diagram of dispersion of light through a triangular glass prism.
[4 marks]
3. (a) (i) What is soil erosion?
(ii) Give two types of soil erosion
[4 marks]
(b) Write the name and chemical formula of the compounds formed when the following elements
combine:
[4 marks]
[7 marks]
4. (a) (i) What is a hormone?
(ii) Copy and complete the following table
Name of hormone Organ secreting
hormone Function of
hormone
Adrenalin
Thyroxin
Insulin
[6 marks]
(b) Ammonia gas is prepared in the laboratory by heating ammonium chloride and calcium hydroxide.
(i) (ii) (iii) Write a balanced equation for the reaction. How is ammonia gas tested in the laboratory? State one use of ammonia.
(c)
(i) (ii)
Name four types of forces.
A boy used a catapult to kill a bird which was on a tree and it fell to the ground. Give two forces involved in this process
[3 marks]
5. (a) (i) Distinguish between a rhizome and a stem tuber
(ii) Give one example each of a rhizome and a stem tuber
[5 marks]
(b) State the method which could be used to separate each of the following mixtures and explain your answer in each case:
(i) salt and water,
(ii) sawdust andiron filings,
(iii) powdered charcoal and water
[6 marks]
(c) (i) Define density of a substance
(ii) Explain how a body which sinks in water could be made to float.
[4 marks]
CLICK TO VIEW ANSWERS TO PART 2
SCIENCE Section B
SOLUTIONS
ESSAY
1. (a) (i) I – Cell
II – key/ switch
III – Ammeter
IV – Electric bulb
V – Variable resistor
VI – Voltmeter
(ii) The key/switch
(iii)
The bulb will light,the ammeter will deflect, the volt meter will deflect
(iv) Chemical→Electrical→light (and heat)
(v)
Parallel connection
(b) (i) (ii) Distillation
Liquid Y – Alcohol
This is because it is more volatile and has a lower boiling point than water.Therefore,it boils
first,leaving the water behind.
(iii)
The thermometer measures the temperature within the flask
The water jacket X cools the gaseous alcohol to condense it back to liquid form
(iv)
Heating and cooling/vapourisation and condensation
(v)
Boiling points of water and alcohol which are clearly different.
(c) (i) A – Starch or carbohydrate
B – fats and oils
C – reducing sugar or simple sugar
(ii) Products of digestion
A B C – glucose
– fatty acids and glycerol
– glucose
(iii)
A B C
– Mouth
– The duodenum(first part of the small intestines)
– Mouth
(iv) C is absorbed after digestion in -The ileum(third & last part of the small intestines
2. (a) (i) Four parts of a plant used for a sexual reproduction
Rhizome, bulb, sucker,stem cutting, tuber, corm, bud [any four]
(ii) Two Advantages of sexual reproduction over a sexual reproduction
Offspring are usually more resistant to diseases
Offspring are usually more stronger and healthier
Offspring have different and usually better characteristics from parents
[anytwo]
(b) (i) Why atoms are electrically neutral
The numbers of protons and electrons are equal.The positively charged protons neutralize
the negatively charged electrons, hence, making the atom electrically neutral
(ii)
Systematic names of:
(α) CuO – Copper(II)Oxide
(β) SO2 – Sulphur (IV)oxide
(c) (i) Dispersion of light
The separation of light into different wave lengths or its component colours when it passes through a given medium
(ii)
Labeled diagram showing dispersion of light through a triangular glass prism

3. (a) (i) Soil erosion
The washing away of the top soil by agents of erosion
Or:
The removal of soil material by natural processes,principally running water, glaciers, waves, andwind
(ii) Two types of soil erosion
Sheet, Gully, Rill, Splash [any two]
(b) (i) Zinc and oxygen
Name: Zinc oxide
Chemical formula: (ZnO)
(ii) Calcium and Chlorine
Name: Calcium chloride
Chemical formula: CaCl2
(c) (i)
Heat Temperature
The energy possessed by a body that
results in its hotness or coldness A measure of the hotness or coldness
of the body
Unit = joule (J) Unit = kelvin (K) or
degee Celsius (°C) or degree Fahrenheit(°F)
(ii) Labeled diagram of mercury-in-glass thermometer
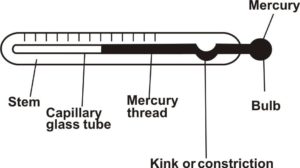
(iii) Mode of heat transfer = Conduction
4. (a) (i) Hormone
A chemical secreted by an endocrine gland or some nerve cells that regulates the function of a specific tissue or organ
(ii)
Name of hormone Organ secreting hormone
Function of hormone
Adrenalin
Adrenal gland increases the speed and
force of the heart beat or
increases the energy of
the body.
Thyroxin
thyroid gland stimulates metabolism
for normal growth and development of the body
Insulin
Pancreas regulates the glucose
(sugar)level in blood
(b) (i) Balanced equation for the laboratory preparation of ammonia
NH4Cl + Ca(OH)2 → NH3 + CaCl2+ H2O
(ii) Test for ammonia gas
Hydrochloric acid is introduced into the gas. White fumes are produced in the presence of the
HCl.
Or: Gas is smelled. Ammonia has a very pungent/choky smell.
(iii) Uses of ammonia
As a refrigerant(used in refrigerators)
Preparation of fertilizers
Flying of balloons
Preparation of medicines/drugs
Preparation of explosives [anyone]
(c) (i) Types of forces
Frictional, tensional, gravitational, centripetal, centrifugal, magnetic,upthrust
[any four]
(ii) Forces involved
Tensional force(in the stretched catapult);
frictional force(bird perched on the tree)
gravitational force (bird falling to the ground); [any two]
5. (a) (i) Differences
Rhizome Stem tuber
Grows horizontally below the ground Grows vertically/diagonally below the
ground
Buds are covered by scaly leaves Buds are not covered by scaly leaves
Both nodes and inter nodes present Only nodes present
(ii) Examples of
Rhizome: Ginger, canna lily
Stem Tuber: Potato, yam
(b) Method of separation and explanation
(i) Salt and water – Evaporation
The temperatures at which water and salt evaporate are wide apart. Therefore, as the mixture is heated, the water evaporates, leaving the salt crystals behind
(ii)
(iii) Saw dust and iron filings – Magnetization
Their on filings is a magnetic substance, while saw dust is not.Hence, the magnet would attract their on filings, leaving the saw dust behind
powdered charcoal and water-Decantation or filtration
Powdered charcoal particles are insoluble in water;therefore they can be separated from water by
filtration or decantation, if the particles have settled at the bottom
(c)
(i)
Density of a substance
The mass perunit volume of the substance
Or: Density = mass /volume
(ii)
Explanation
When the volume of a body increases at a constant mass, its density reduces.Therefore,the
volume of the body is increased by increasing its hollowness/expanse such that its resultant
density is less than that of water.Then the body will float on water.